Реакции циклических оксидов в условиях бифункционального гетерогенного кислотно-основного катализа
Реакции циклических оксидов в условиях бифункционального гетерогенного кислотно-основного катализа
Аннотация
Реакции циклических эпоксидов составляют основу многих промышленных химико-технологических процессов. В отличие от простых эфиров, оксираны склонны подвергаться реакциям нуклеофильного расщепления. Химические свойства циклических алканов в первую очередь определяются размером их колец. Искажение тетраэдрических углов в малых кольцах приводит к изменению химических свойств веществ. Химические свойства оксиранов существенно отличаются от свойств простых эфиров, устойчивых к расщеплению в обычных условиях. Как и циклопропан, оксиран легко раскрывается в мягких условиях и служит основой для синтеза различных производных. Обычно небольшие кольца характеризуются реакциями присоединения после расщепления кольца. Однако в кольцевых молекулах, содержащих пять и более атомов углерода, валентные углы близки к тетраэдрическим, что делает их устойчивыми к расщеплению и более характерными для реакций замещения, чем присоединения.
1. Introduction
One of the promising areas of chemical technology is processes using cyclic oxides with bifunctional heterogeneous acid-base catalysis.
In a work published in 2003, it was noted that 103 processes using solid acids, 10 using solid bases, and only 14 processes using bifunctional acid-base catalysis were implemented in industry. Today, tens of times more such processes have been implemented .
2. Main Part
One of the most promising bifunctional catalysts are layered double hydroxides , , , .
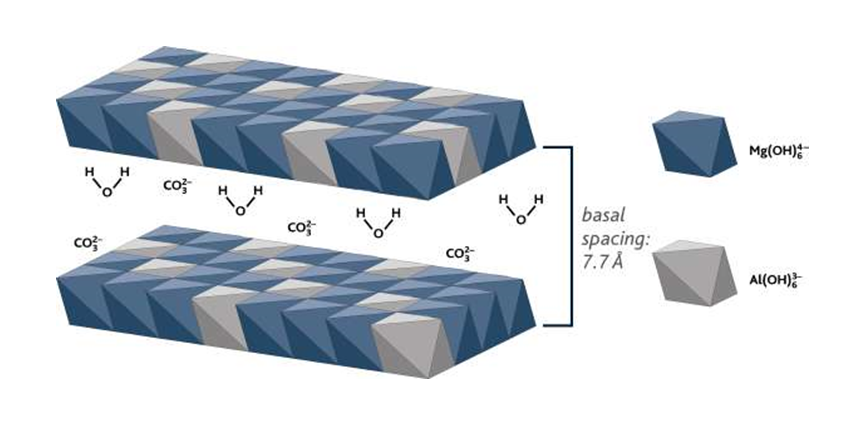
Figure 1 - Structure of layered double hydroxides
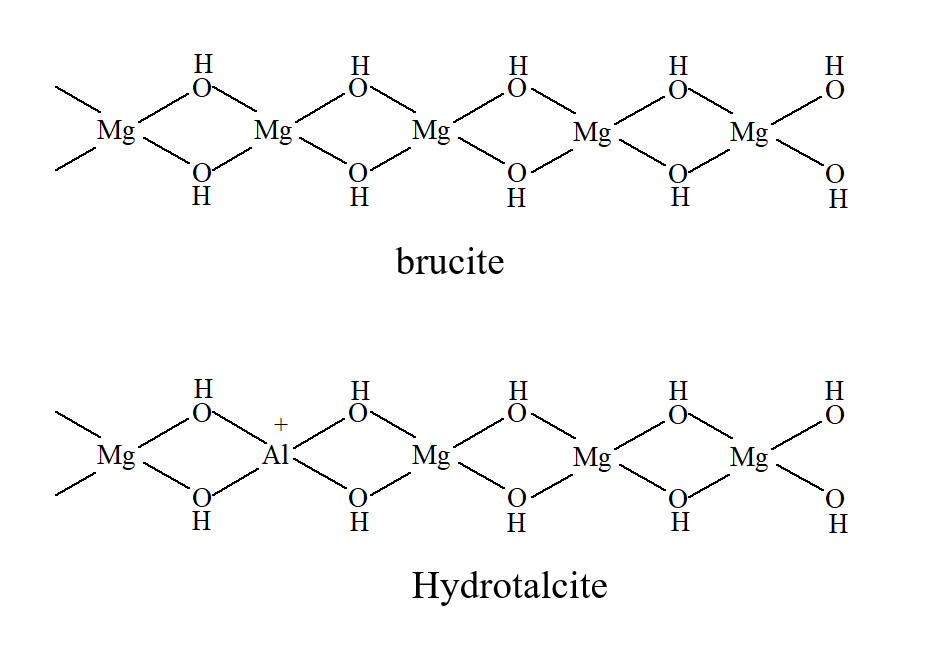
Figure 2 - Example
Therefore, it is always possible to calculate the degree of isomorphic substitution leading to the maximum number of basic sites on the catalyst surface.
Often this degree of substitution corresponds to the most stable catalyst structure. The lowest strength of the main sites is possessed by hydroxide catalysts constructed from metals of equal oxidation states. Their strength is determined only by the inductive effect due to the shift in electron density.
The Brønsted site in such compounds can be a hydroxyl localized on tetrahedral aluminum. Lewis ground sites represent the lone electron pair of oxygen.
Since the number of active sites depends on the degree of isomorphic substitution, a double hydroxide of two metals with different oxidation states can be either a basic compound or one with acidic sites.
There is an inverse relationship between strength and the number of main sites. That is, the center of the sample in which one single atom is isomorphically replaced will have the maximum strength; the capacity of such a sample will be minimal. Accordingly, the sample with the maximum number of main sites will have the minimum strength of main sites.
At any Me(N)/Me(M) ratio, the actual structure of complex hydroxide catalysts will be far from ideal, that is, from a uniform alternation of isomorphic substitution sites. During syntheses, especially hydrothermal ones, there is no substitution, but rather an increase in the oxide chain during the synthesis process, and the sequence of cations is determined statistically, considering their reactivity. If the growth of the oxide chain is determined statistically, then real complex hydroxides, at almost any stoichiometric ratio, have acid-type sites, although their acidity can be practically unnoticeable given the strong basic properties of the elements forming the hydroxide layer.
Of course, the number and strength of acid-type sites also depends on the degree of isomorphic substitution. When studying the basicity and acidity of catalysts with different ratios of cations, an extreme dependence on composition was established (Table 1). For the given catalyst, the main sites lie in the region 17.2 < H0 < 18.
Table 1 - Changes in the acid-base properties of double hydroxide with changes in composition
MgO/Al2O3, mol/mol | 2,49 | 3.80 | 5,23 | 9,33 | 17,6 |
Acidity, meq/g | 0,41 | 0,32 | 0,21 | 0,06 | 0 |
Basicity, meq /г | 0,73 | 0,54 | 0,63 | 0,85 | 0,94 |
LDHs can find the widest application in reactions with cyclic oxides, since such processes most often occur with the participation of a center of a different nature.
The review provides data on the use of LDH in reactions such as addition to C=O, C=C and С=С, C=N bonds, alkylation, acylation, decarboxylation, as well as reactions of epoxides. Similar results are described in publication .
Let us consider examples and features of the reaction of cyclic oxides on bifunctional catalysts, for example, oxyethylation of 1-dodecanol on a hydrotalcite-like compound pre-treated with dodecanoic acid. Treatment with dodecanoic acid leads to a significant increase in catalyst activity while maintaining a narrow distribution of products .
The narrow distribution of products can be explained by the following. Relatively basic substances, ethylene oxide and alcohol, are sorbed on acidic Al3+ sites. Ethylene oxide adsorbed on neighboring sites prevents reaction products from being sorbed on neighboring sites. The rate of sequential oxyethylation decreases due to the small number of sites with adsorbed ethylene oxide near the sites on which the reaction products were adsorbed. The activity of hydrotalcite depends on the Mg-Al ratio, which determines the distance between two adjacent Al3+ sites.
A comparison of the rates of dodecanol oxyethylation catalyzed by hydrotalcite and NaOCH3 showed that at the initial time the rate of the heterogeneous process is significantly lower, but the total rate with homogeneous catalysis is almost twice as high. This may be due to the irreversible adsorption of reaction products on active sites.
The distribution of dodecanol oxyethylation products (mcat=0,5 %, Т=180 оС) in the presence of various catalysts was studied. MgAl-hydrotalcite gives a narrower distribution of products compared to NaOCH3.
When oxyethylation of 1-butanol on hydrotalcite-type catalysts, into which a small amount of KOH is introduced, gives a ratio of mono- and di-addition products greater than 10:1. This can be explained by the fact that there is competition for the active centers of alcohol molecules, which are more basic, with product molecules.
Hydrotalcites intercalated with various polyoxometalates, such as chromates, dichromates or vanadates, give a monoderivative with 100% selectivity during the oxyethylation of 1-butanol.
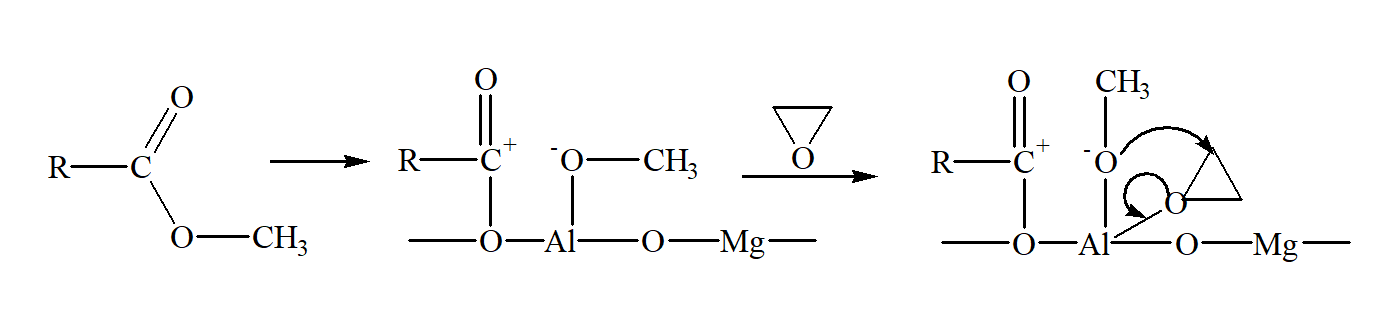
Figure 3 - Reaction mechanism
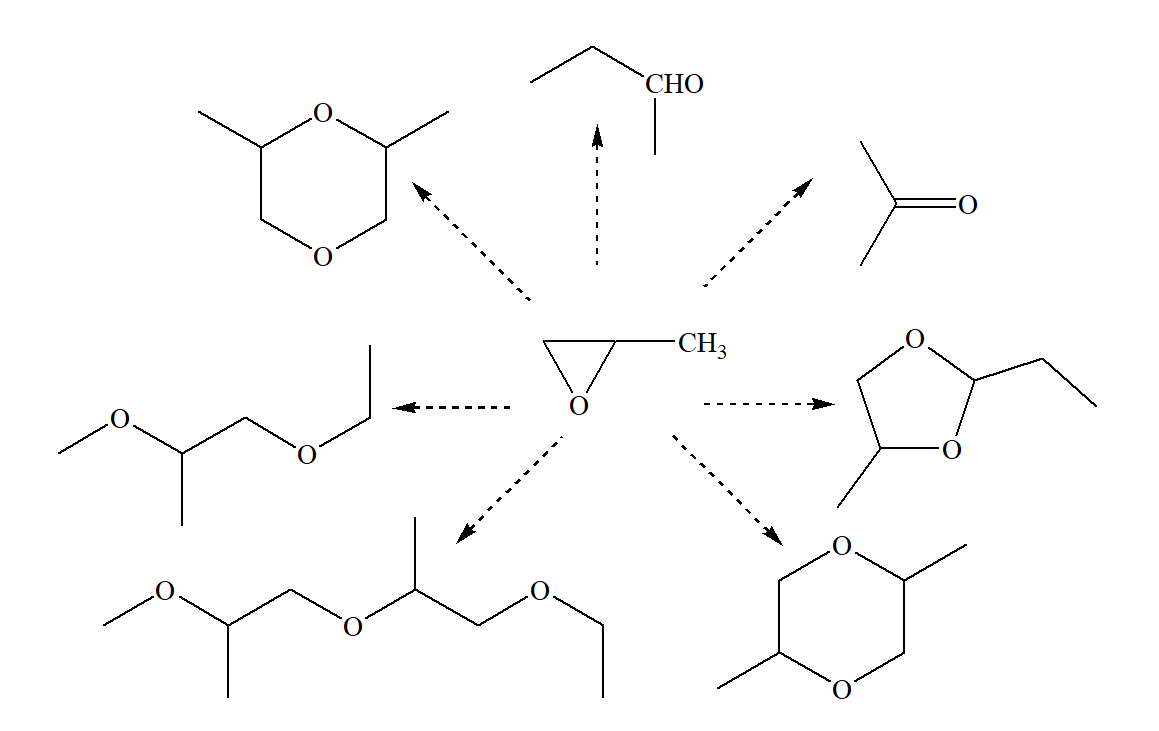
Figure 4 - Products
In the presence of solid acids with relatively strong sites, all products are formed except acetone. Most of all, cyclic dimers are formed, as well as propylaldehyde. In addition, linear polymerization products are found in the reaction products, which are not formed when zeolites are used.
A less strong acid is montmorillonite – the main product is propylaldehyde, as well as 1,4-dioxane.
When catalyzed by Mg-Al-hydrotalcite, the main catalyst, the reaction products are propylaldehyde, acetone, and polyaddition products.
In the presence of Na-MCM-41 zeolite, which has strong basic and acidic sites, cyclic dimers predominate among the products, and polyaddition products are absent, which indicates that in this reaction the rates of acid-catalytic processes are much higher than the reactions on basic catalysts.
It is known that if a reaction with an ethylene oxide derivative begins with an acid attack, a whole spectrum of products is formed, in contrast to a base attack, which leads to the formation of a single product. This suggests that both basic and acidic sites operate on these catalysts. In acid heterogeneous catalysis, both SN1 and SN2 reaction mechanisms are realized, and in basic catalysis, only SN2. The SN1 mechanism leads to the formation of propylaldehyde, and SN2, in both acid and base catalysis, leads to the formation of acetone.
The reaction of lauric acid with ethylene oxide with a molar ratio of 1:3, at a temperature of 130-135 oC was studied . The homogeneous process was carried out in the presence of catalysts such as potassium isobutoxide, which showed better activity and selectivity than potassium methylate, ethoxide and butoxide.
Hydrotalcite impregnated with potassium isobutoxide was studied as a heterogeneous catalyst, which showed lower activity than a similar homogeneous one, but with higher selectivity. The conversion of ethylene oxide after 3 hours of reaction is at 3% catalysis content in the reaction mixture: with homogeneous catalysis – 80%, with an impregnated catalyst – 50%, in the presence of untreated hydrotalcite – less than 1%.
The following were found in the reaction products: diethylene and triethylene glycols; mono, di- and tri- and tetraoxyethylated products; as well as diesters of mono-, di- and triethylene glycol.
The hydration reaction of ethylene oxide on the binary oxide Nb2O5/a-Al2O3 proceeds at a significantly higher rate , than on zeolites or aluminophosphates, and is comparable to the rate of pure niobic acid Nb2O5 . nH2O, and with much higher selectivity for monoethylene glycol.
A similar catalyst, with acidic and basic sites, CsF supported on selitol, showed high activity in the reaction of epoxides with thiol. The reaction occurs under mild conditions, with the selective formation of β-hydroxosulfides.
When studying ring opening in the presence of catalysts HZSM-5, CuZSM-5, HY, AlMCM-41, NaN3-AlMCM-41, the occurrence of three types of reactions was established: dimerization, isomerization, and reaction with CO2 elimination . At moderate temperatures, only the first two reactions occur, with acid catalysis, since in the presence of NaN3-AlMCM-41 no changes occur.
The isomerization reaction of styrene oxide with the formation of b-phenylacetaldehyde occurs in the presence of many basic heterogeneous catalysts – hydrotalcites, binary oxides, rare earth metal phosphates, KF on a carrier .
Despite the high activity of the catalysts, they are quickly deactivated by polymerization products. ZnO/Al2O3 exhibits the highest activity with 100% selectivity for b-phenylacetaldehyde.
During the oxyethylation of alcohols catalyzed by heteropolyacids, active formation of ethylene glycol diesters occurs.
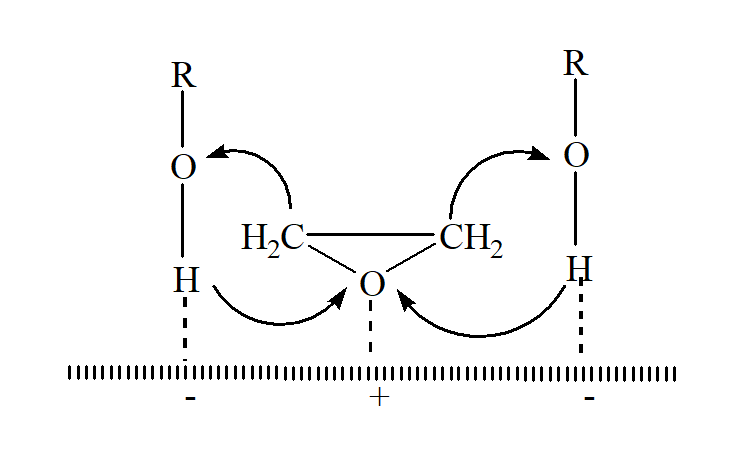
Figure 5 - Process diagram
The reactions of propylene oxide with methanol in the presence of MgO and CaO were studied . Magnesium oxide has basic sites of moderate strength and is significantly more active than calcium oxide, which has stronger basic sites. It was found that methanol dissociates on the MgO surface to form methoxyl anions, and weak Lewis acid sites also take part in the reaction.
The reactions of CO2 with cyclic oxides were studied. When using MgO and Mg-Al oxide obtained by calcination of hydrotalcite as catalysts, stereospecific addition to the oxide was discovered.
For reactions of CO2 with ethylene oxide and epoxypropylbenzene catalyzed by basic zeolites, anion exchangers, Cs/Al2O3, Cs/MgO, it was shown that the reaction rate increases in proportion to the basicity of the catalyst.
With homogeneous catalysis, the rates of addition of CO2 to ethylene oxide and to epoxypropylbenzene are approximately equal. In heterogeneous catalysis, due to the sieve effect, when using zeolites X and Y, the reaction with epoxypropylbenzene does not occur, and when using non-porous MgO, the rates are also equal.
The reaction rate for catalysis of Mg-Al oxides is higher than for MgO due to the presence of Lewis acid sites.
The reaction mechanism is as follows. CO2 is adsorbed on the main Lewis sites (O2-), forming surface carbonates, so the strength of the sites determines the course of the entire process. Ethylene oxide is sorbed at the nearby Lewis acid site. The surface carbonate opens stereospecifically, reacting with the carbon atom of the adsorbed oxirane to produce an oxyanion. In basic zeolites, the ion-exchange cation provides a coordination site for oxiranes.
3. Conclusion
Reactions of cyclic oxides are important for industrial processes. The chemical properties of cycloalkanes depend on the size of the cycle.
The strong difference from the tetrahedral angle in small cycles determines the chemical properties of the molecules.
Oxirane, the simplest cyclic ether, is an outstanding exception to the generalization that most ethers are resistant to cleavage. Like cyclopropane, the three-membered ring is highly strained and readily opens under mild conditions. Indeed, the importance of oxуcyclopropane as an industrial chemical lies in its readiness to form other important compounds.
